Neural Underpinning of Cognitive and Behavioral Deficits in Psychotic Spectrum Disorders
Overview
We are working to uncover the mechanisms of psychotic spectrum disorders and develop biomarkers for gauging disease progression and response to treatment.
Psychotic Spectrum Disorders (PSD), which include schizophrenia, schizoaffective disorders, and mood disorders with psychotic features, are complex conditions of unclear etiology, variable disease course, and broad range of outcomes. Current medications address only in part the behavioral and cognitive deficits characteristic of PSD. We are pursuing a multi-modal approach focused on 1) revealing the neural mechanisms that underlie the behavioral and cognitive deficits with the goal of informing the development of more efficient therapies and 2) providing objective biomarkers for assessing disease progression and treatment efficacy.
Keywords
- Myelin
- Perfusion
- Iron
- Schizophrenia Spectrum Disorders
- Cognition
Project Team
External Collaborators
- Ryn Flaherty, New York University
- Yiyan Lin (NTV)
- Hayden Redfield (NTV), NYU Langone Health
- Sarah Alzarooni (NTV), NYU Grossman School of Medicine
- Donald C. Goff, MD , NYU Langone Health
- William Gordon Frankle, MD, NYU Langone Health
Publications
- McKenna FF, Miles L, Babb JS, Goff DC, Lazar M. Diffusion kurtosis imaging of gray matter in schizophrenia. Cortex. 2019;121:201-224. doi:10.1016/j.cortex.2019.08.013
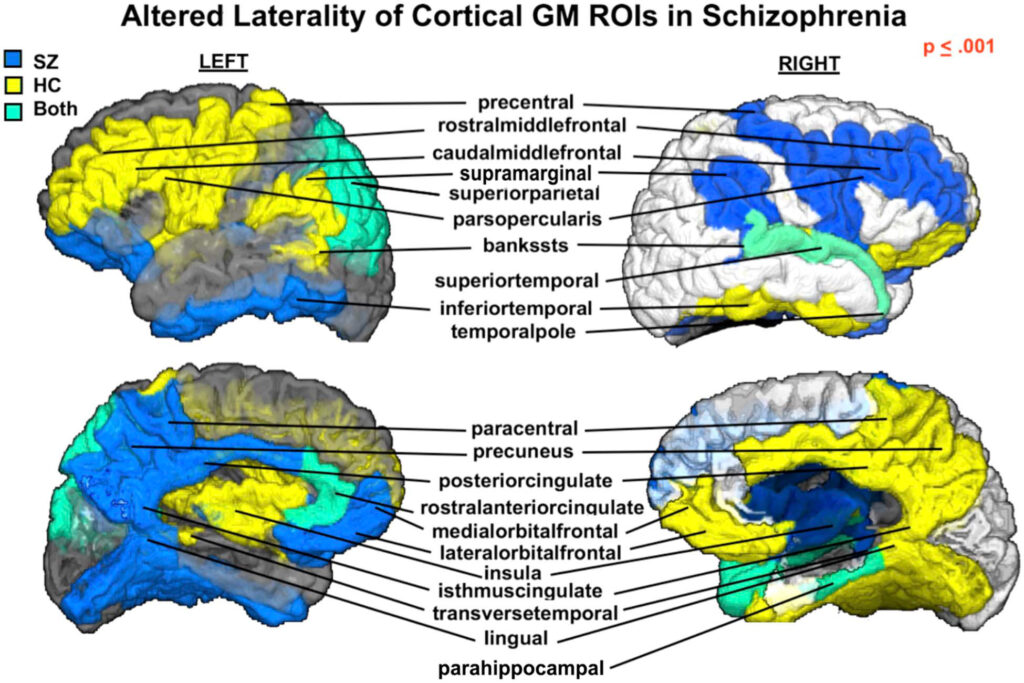
- McKenna F, Babb J, Miles L, Goff D, Lazar M. Reduced Microstructural Lateralization in Males with Chronic Schizophrenia: A Diffusional Kurtosis Imaging Study. Cereb Cortex. 2020;30(4):2281-2294. doi:10.1093/cercor/bhz239
- Sui YV, Bertisch H, Lee HH, Storey P, Babb JS, Goff DC, Samsonov A, Lazar M. Quantitative macromolecular proton fraction mapping reveals altered cortical myelin profile in schizophrenia spectrum disorders. Cereb Cortex Commun. 2021;2:tgab015. doi:10.1093/texcom/tgab015
- Sui YV, McKenna F, Bertisch H, et al. Decreased basal ganglia and thalamic iron in early psychotic spectrum disorders are associated with increased psychotic and schizotypal symptoms. Mol Psychiatry. 2022;27(12):5144-5153. doi:10.1038/s41380-022-01740-2
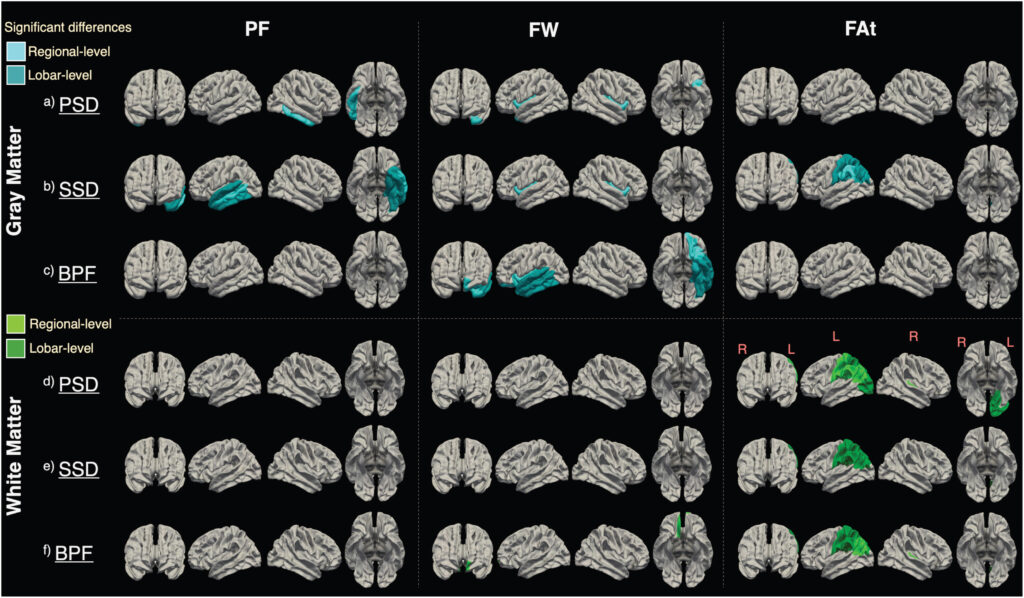
- McKenna F, Gupta PK, Sui YV, et al. Microstructural and Microvascular Alterations in Psychotic Spectrum Disorders: A Three-Compartment Intravoxel Incoherent Imaging and Free Water Model. Schizophr Bull. 2023;49(6):1542-1553. doi:10.1093/schbul/sbad019
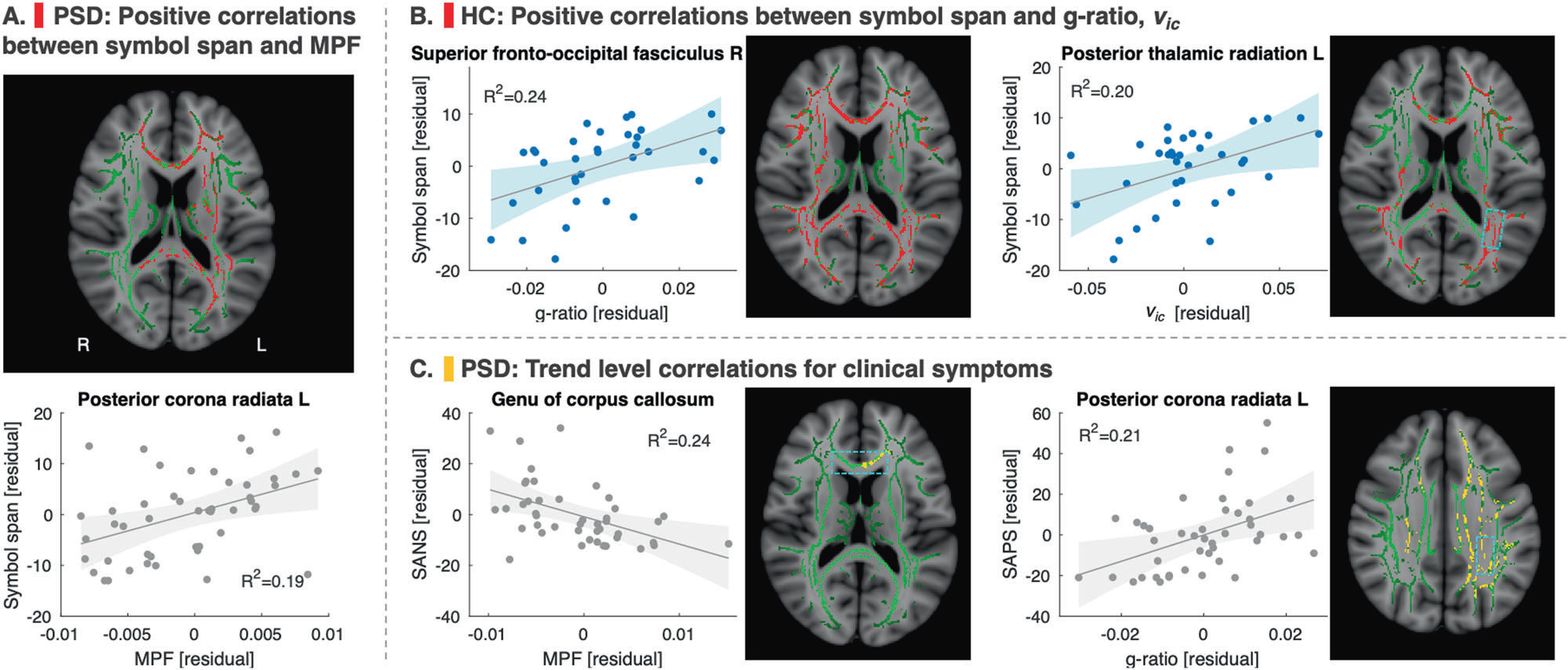
- Sui YV, Bertisch H, Goff DC, Samsonov A, Lazar M. Quantitative magnetization transfer and g-ratio imaging of white matter myelin in early psychotic spectrum disorders. Mol Psychiatry. 2025;30(6):2739-2747. doi:10.1038/s41380-024-02883-0
Acknowledgements
We acknowledge support from the following grants:R21MH085228, R01MH129934, R01MH108962.






